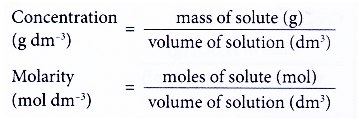How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com

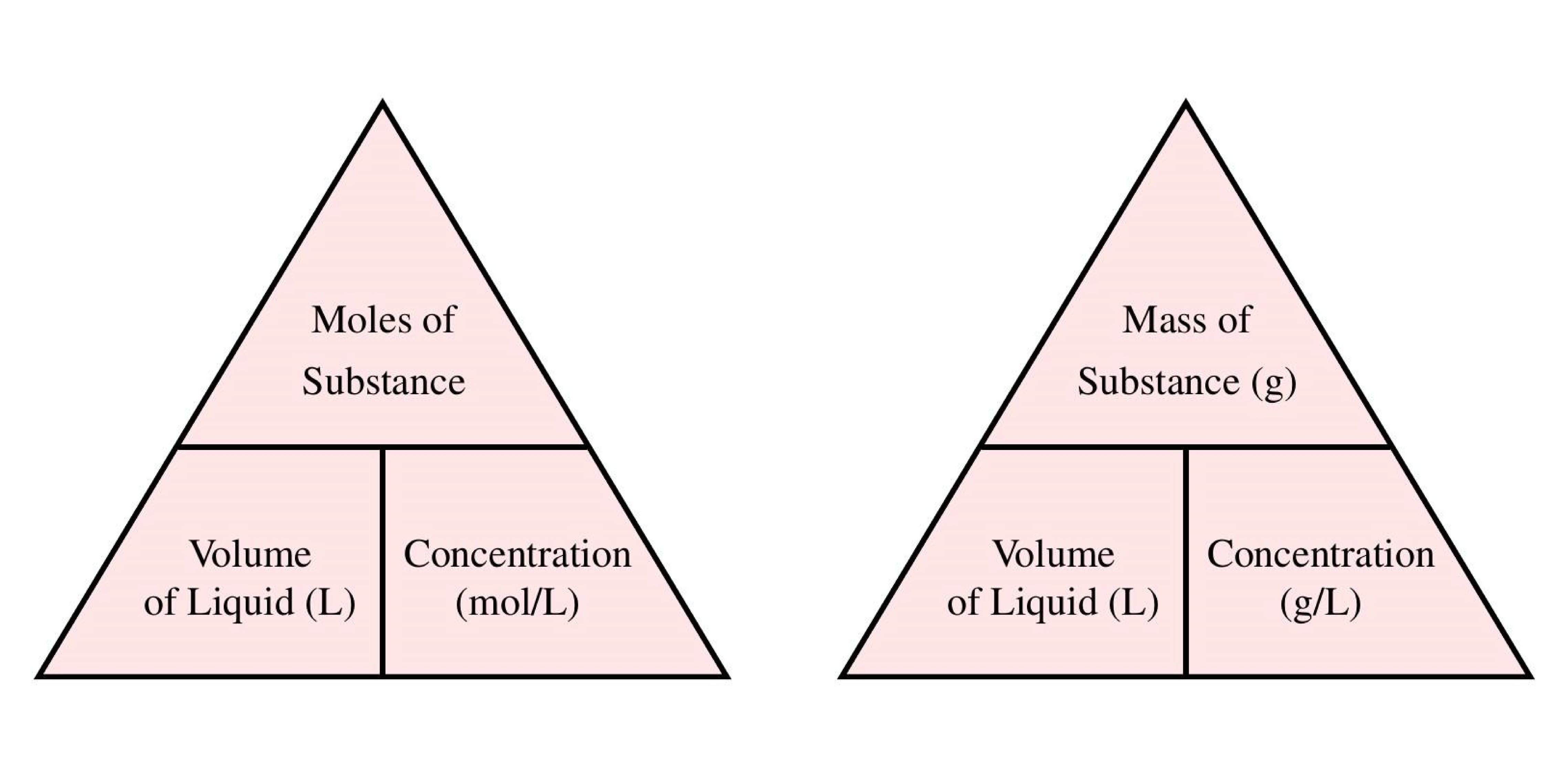
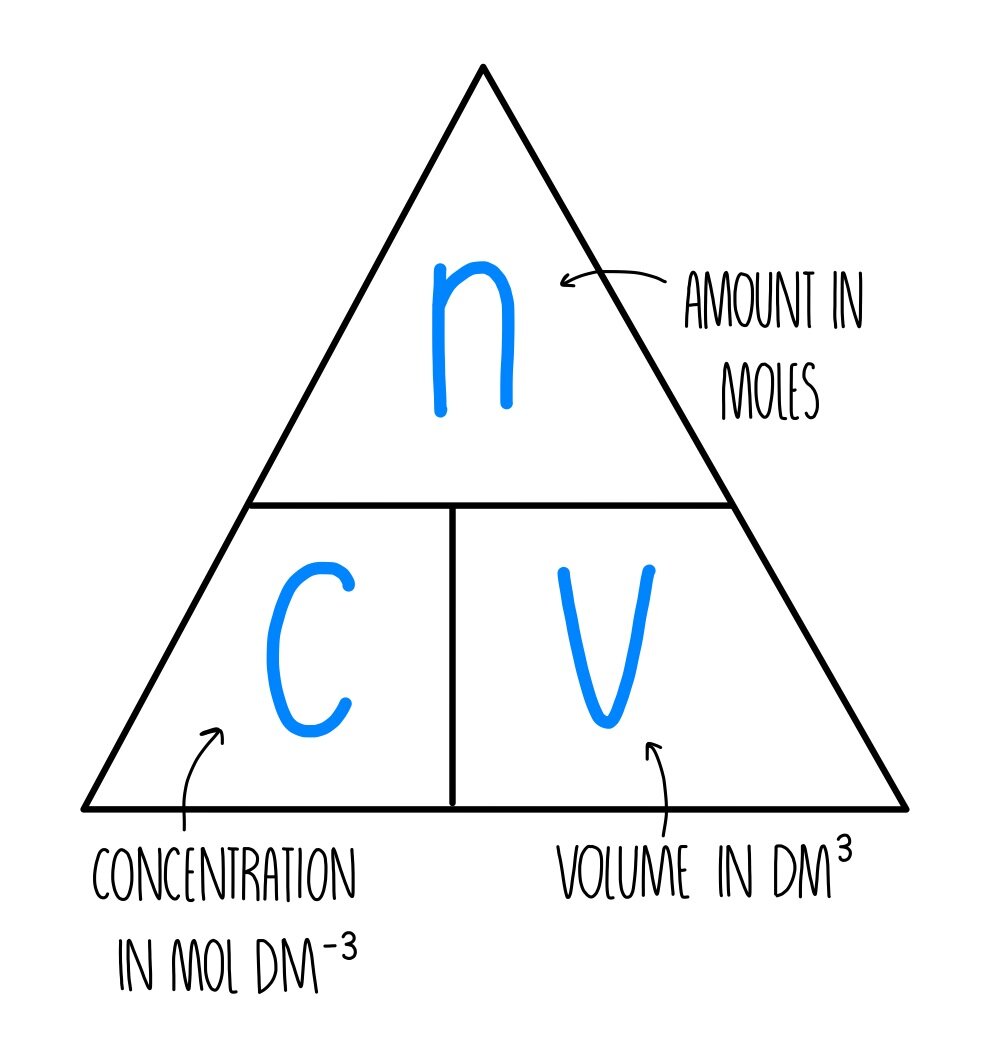
Calculate the concentration interms of mass by volume pecentage of the solution containing 2.5g potassium chloride in 50 ml of potassium chloride (KCl) solution? (AS(1))