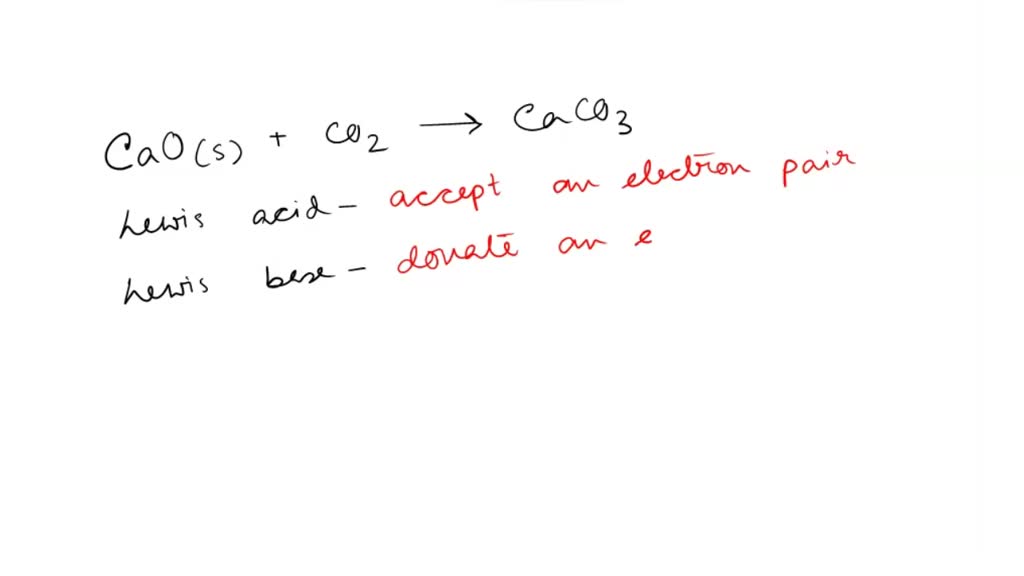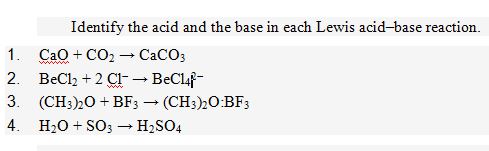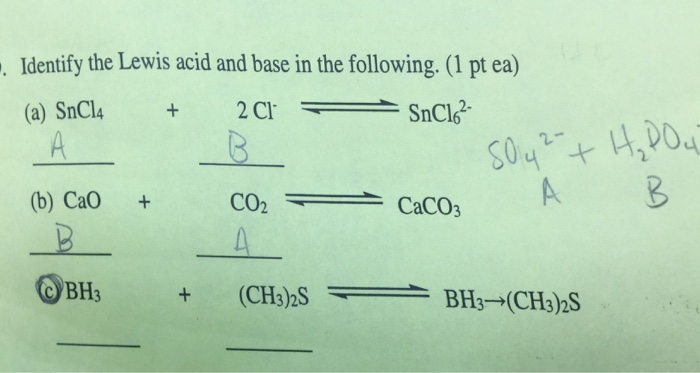SOLVED: The effectiveness of a liming material depends on the chemical compounds that are present: How much each of burned lime and hydrated lime would be equivalent to one ton of calcic

Preparation and application of binary acid–base CaO–La2O3 catalyst for biodiesel production - ScienceDirect
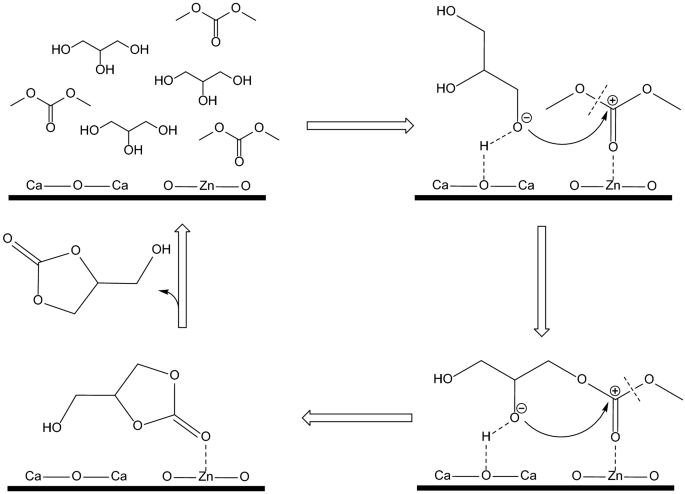
Acidic–Basic Bifunctional Magnetic Mesoporous CoFe2O4@(CaO–ZnO) for the Synthesis of Glycerol Carbonate | SpringerLink

Introduction to Acids and Bases. Acid A substance that produces hydrogen ions, H + (aq), when it dissolves in water. Sour-tasting and good conductors. - ppt download

Magnetic solid base catalyst CaO/CoFe2O4 for biodiesel production: Influence of basicity and wettability of the catalyst in catalytic performance - ScienceDirect

Basic concept of calcium looping gasification with CaO as bed material. | Download Scientific Diagram

A review on the utilization of calcium oxide as a base catalyst in biodiesel production - ScienceDirect
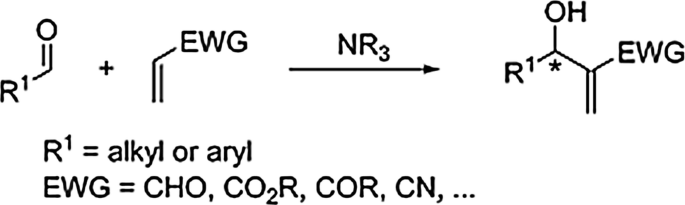
Designed model for the Morita–Baylis–Hillman reaction mechanism in the presence of CaO and CaO modified with ionic liquid as a solid base catalyst: a DFT and MP2 investigation | SpringerLink

Relative Lewis acid-base potential on an arbitrary scale within each... | Download Scientific Diagram

Question Video: Identifying a Feature of the Reactivity of a Carboxylic Acid That Does Not Indicate It Is Acidic | Nagwa
Lynkoa intègre la plus importante base de données de fournisseurs de pièces standards avec fichiers CAO - Visiativ Solutions

SOLVED: Which statement is true for this reaction? CaO + SiO2 CaSiO3 CaO acts as Lewis acid SiO2 acts as a Lewis acid CaSiO3 acts as a Lewis base Si4+ acts as