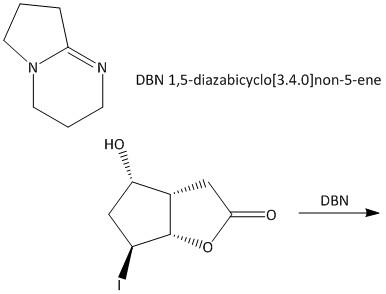
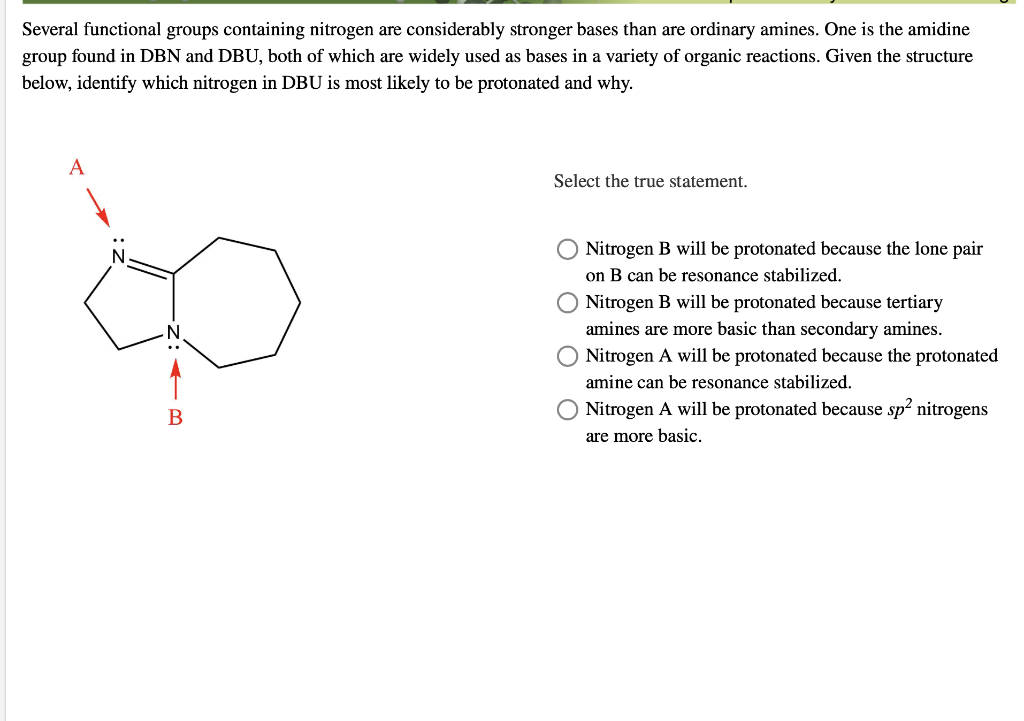
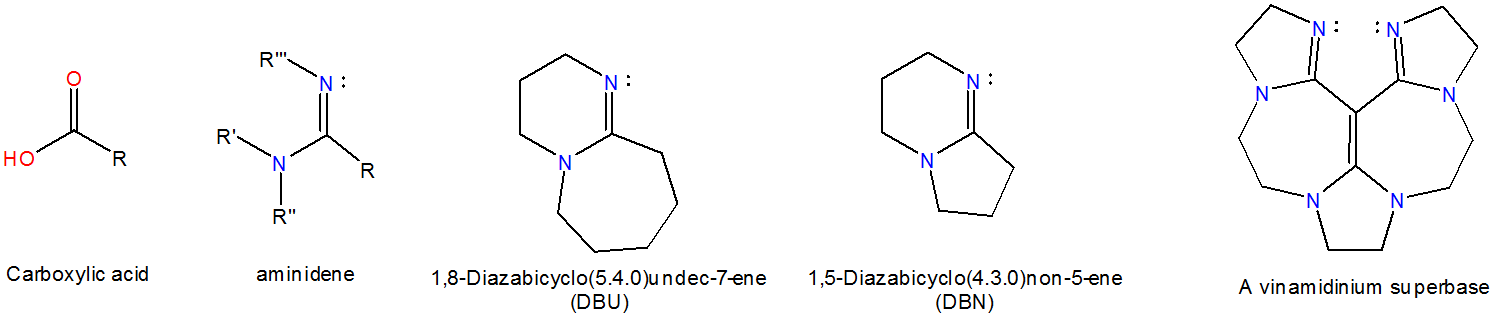
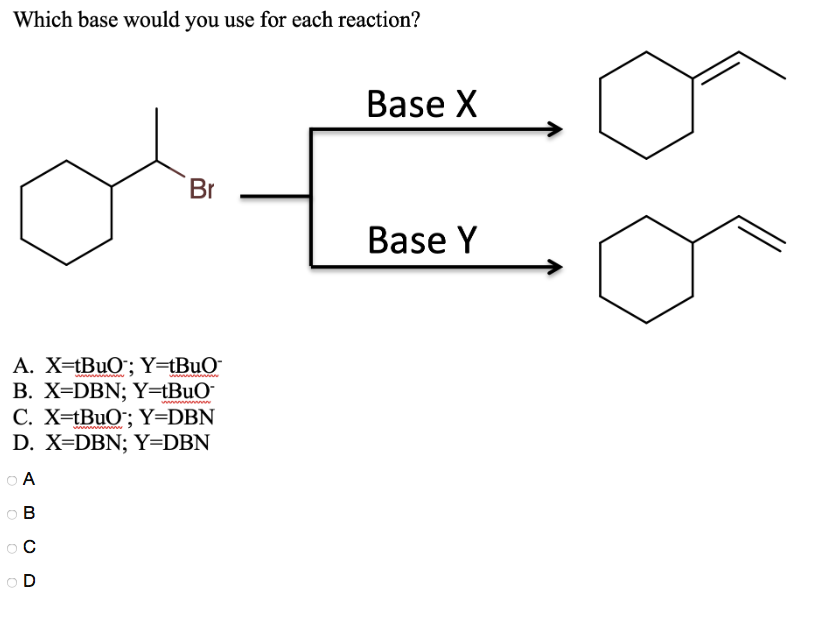
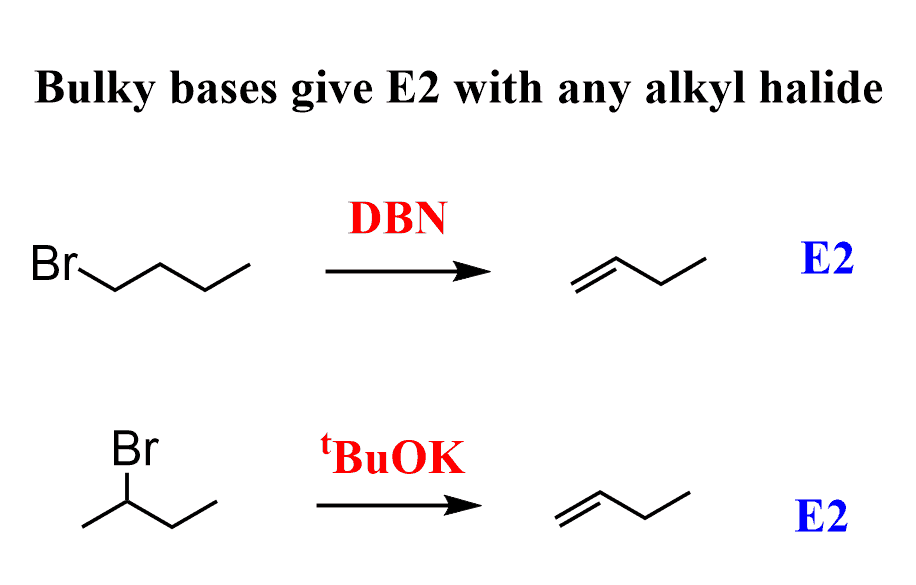
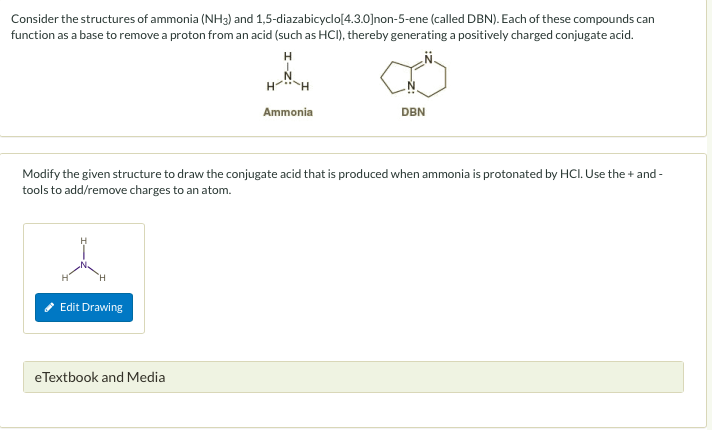
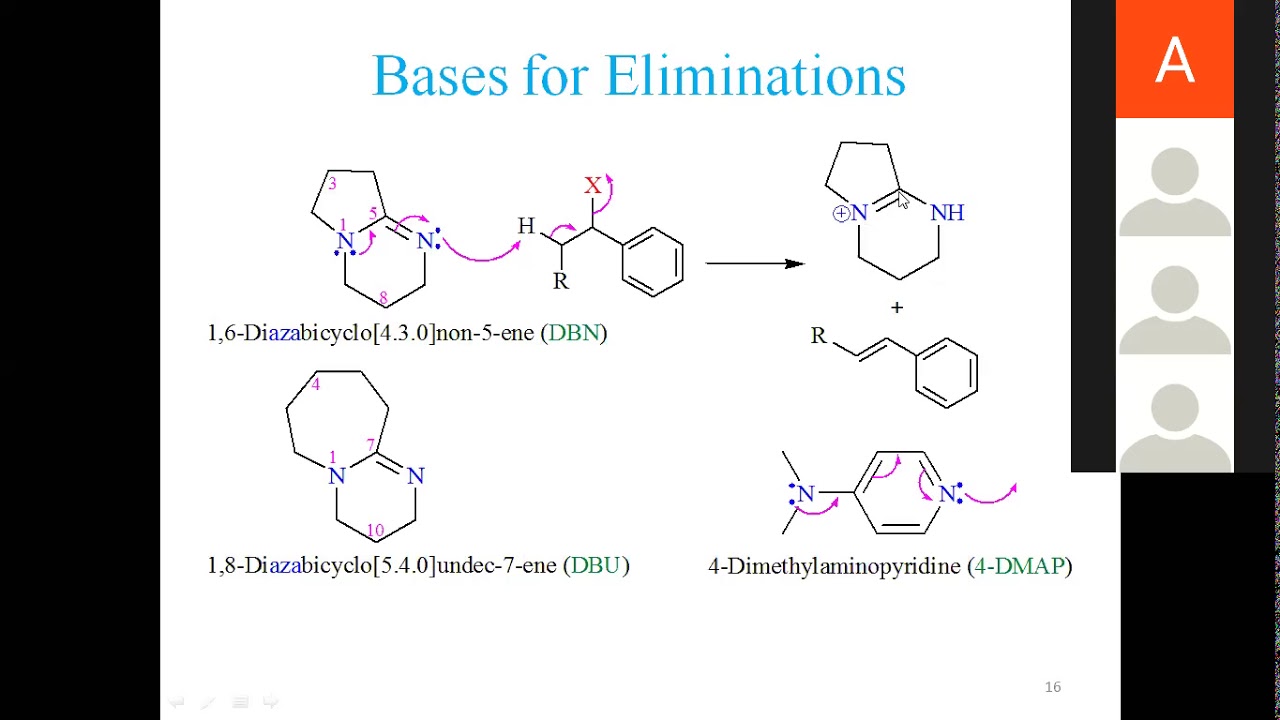
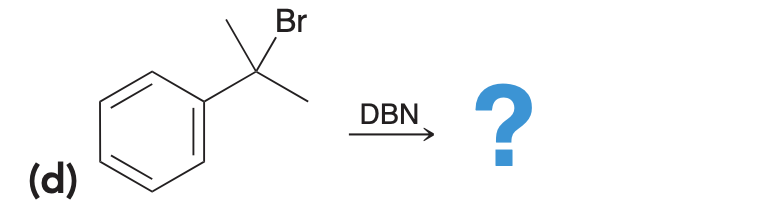
DBN is a bicyclic compound which is used as base. What is the major product in the following reaction?

The basic structures of the restricted Boltzmann machine and the DBN model. | Download Scientific Diagram
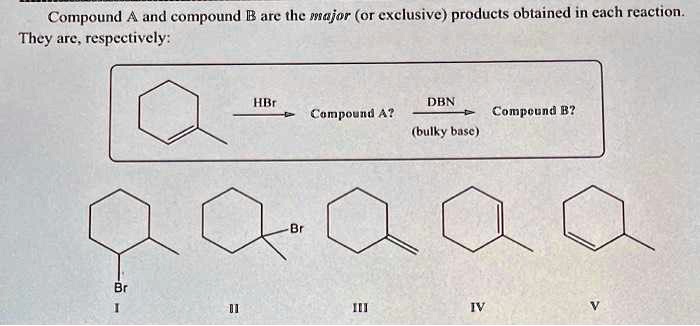
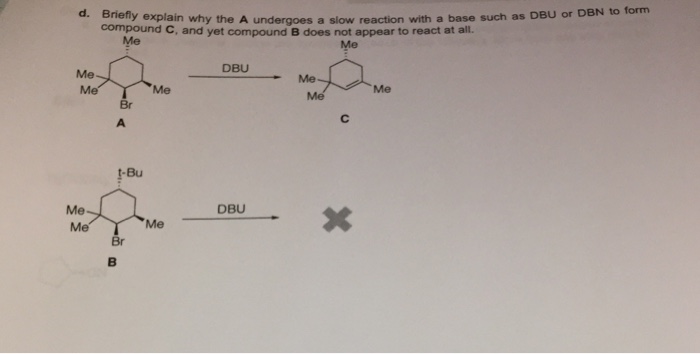
SOLVED: Compound and compound B are the wajor (Or exclusive) products obtained in cach reaction They are. respeetively: HIr DBN Campoyud A? Compcund B? (bulky busc )
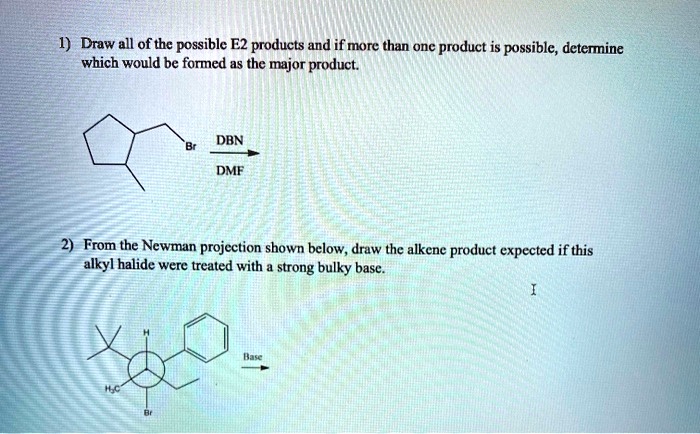
SOLVED: Draw all of the possible E2 products and if more than one product is possible; determine which would be formed as the major product: DBN DMF From the Newman projection shown

GDM in the form of a DBN. The solid lines define the basic structure;... | Download Scientific Diagram
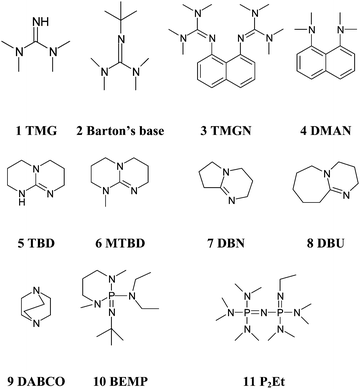
Kinetics screening of the N -alkylation of organic superbases using a continuous flow microfluidic device: basicity versus nucleophilicity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB25215E
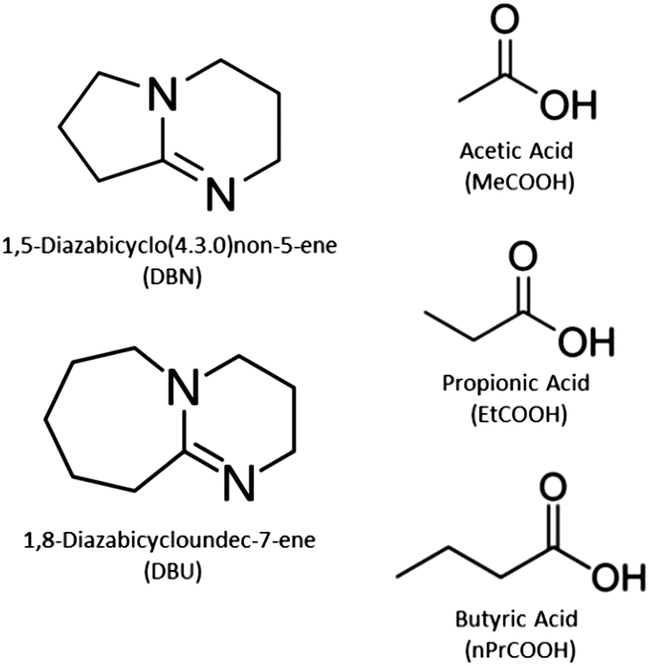
Vaporization of protic ionic liquids derived from organic superbases and short carboxylic acids - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C7CP02023F