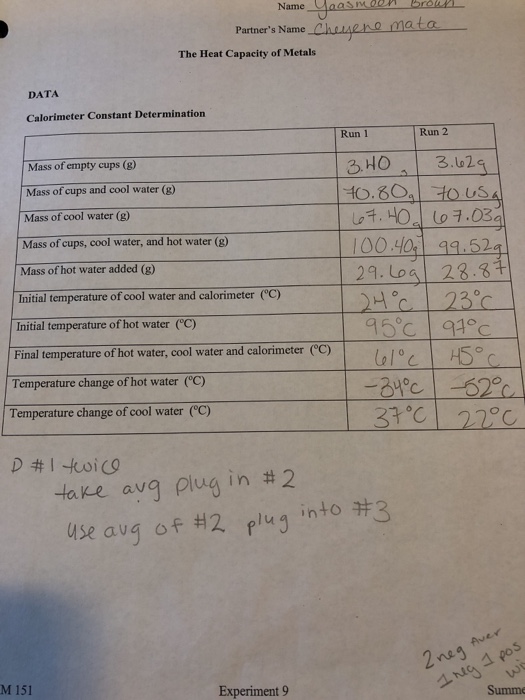
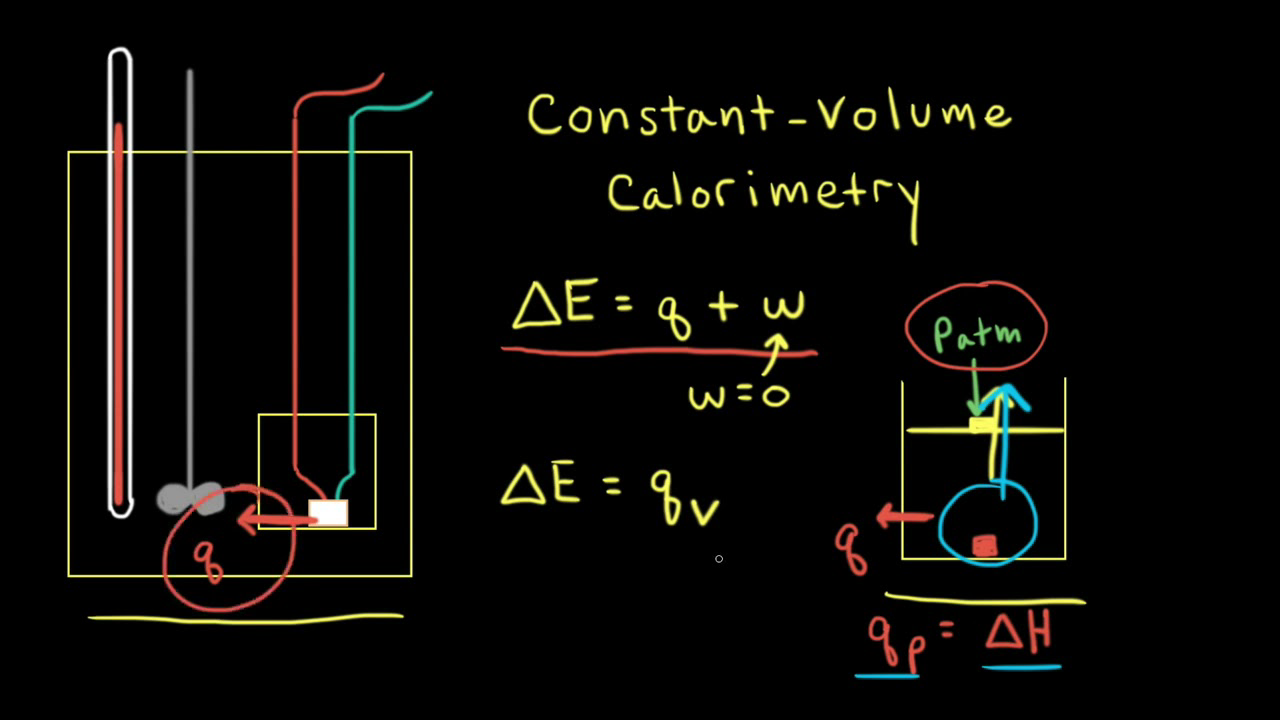
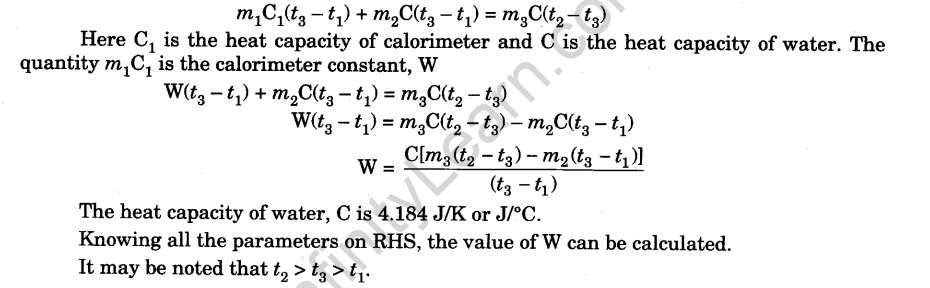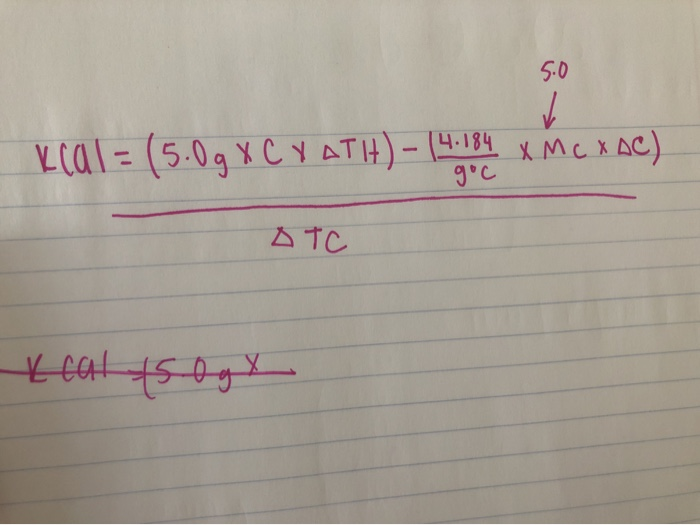When 120 mg of naphthalene C10H8(s) was burnt in a bomb calorimeter, the temperature rise was 3.05 K . Calculate the calorimeter constant and molar standard internal energy change of combustion. Also

Calorimetry Calorimetry is used to measure heat capacity and specific heats. calorimeter: an instrument that measures heat changes for physical and chemical. - ppt video online download

SOLVED: A coffee cup calorimeter contains water at an initial temperature of 20°C and is calculated to have a calorimeter constant (heat capacity) of 75 J/°C. A 28 g piece of an

SOLVED: At constant volume, the heat of combustion of a particular compound is −3833.0 kJ/mol.−3833.0 kJ/mol. When 1.763 g1.763 g of this compound (molar mass=111.48 g/mol)(molar mass=111.48 g/mol) was burned in a