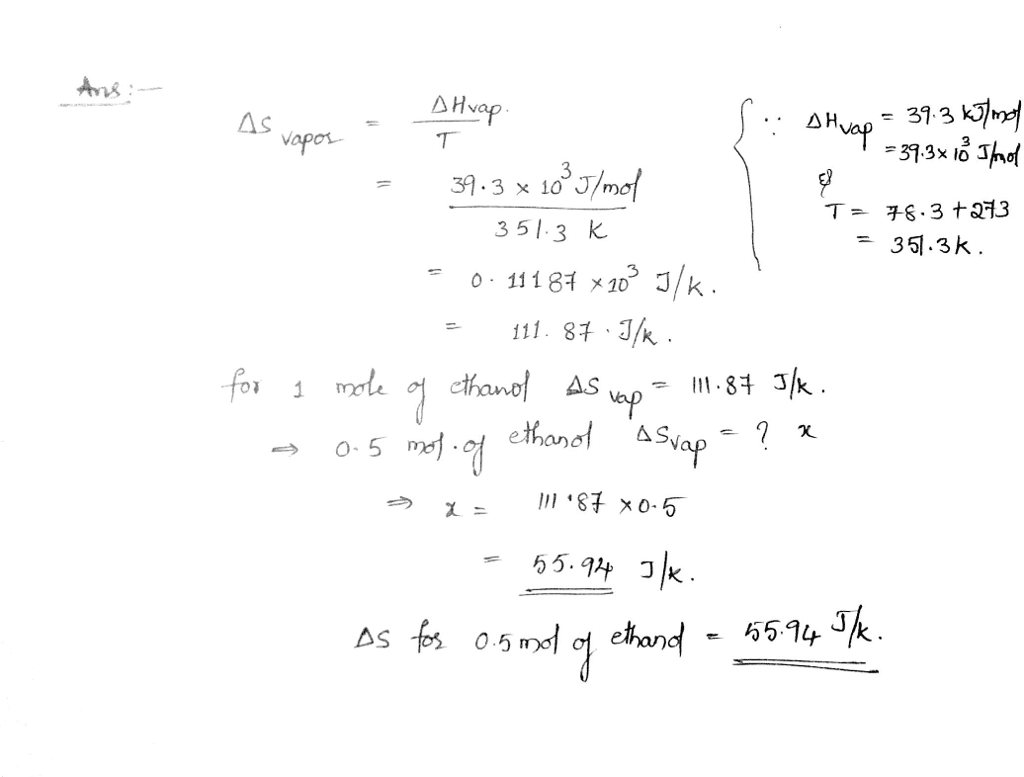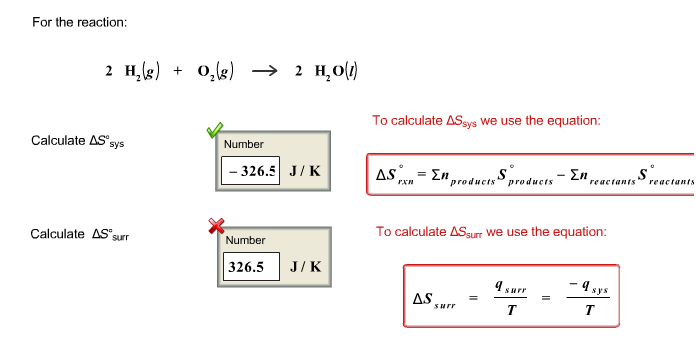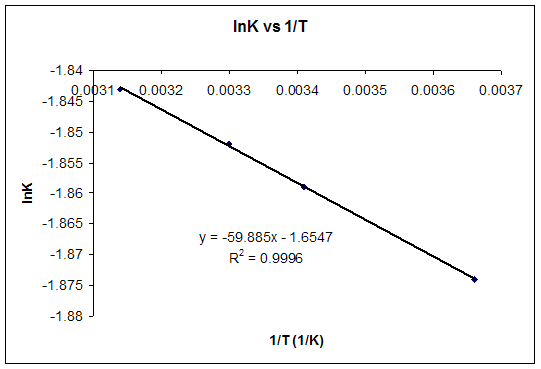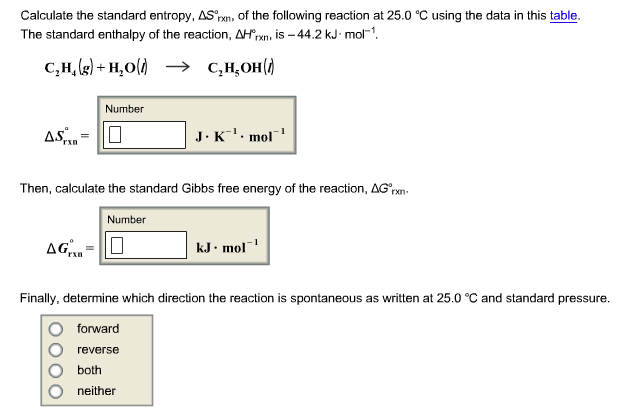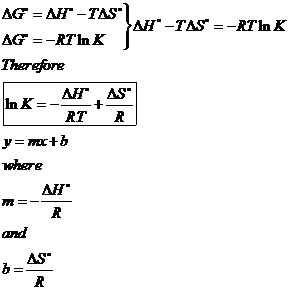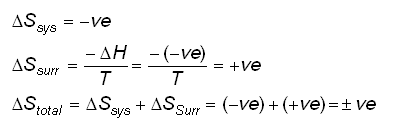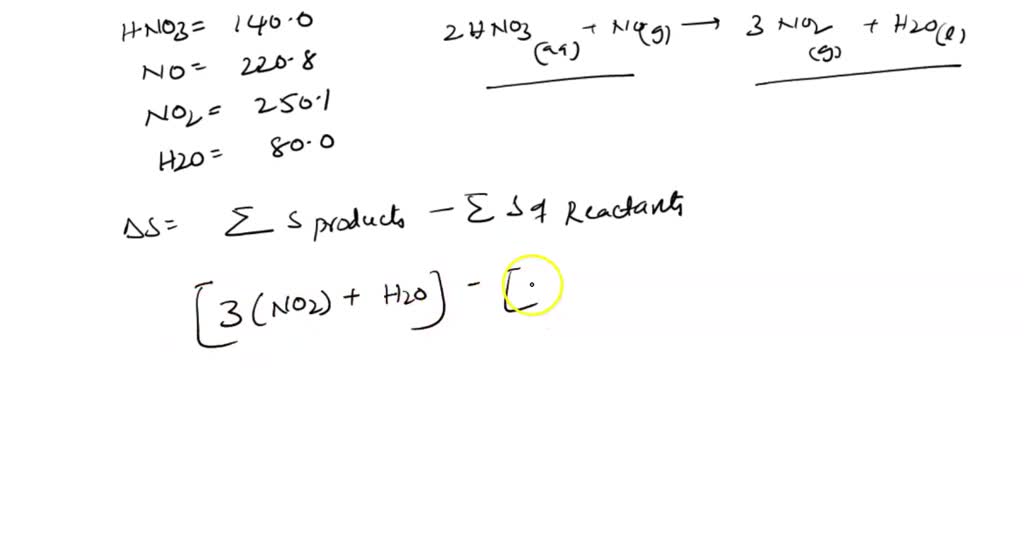
SOLVED: calculate delta S reaction (in J/mol *K) 2HNO3(aq)+NO(g) —> 3NO2(g)+H2O(l) delta S of... HNO3 = 140.0 NO= 220.8 NO2= 250.1 H2O= 80.0

Calculate ΔS univ (in J/K) for the chemical reaction: C(graphite) + 2H2(g)→CH4(g);ΔH300^o = - 75.0kJ . The standard entropies of C(graphite),H2(g),CH4(g) are 6.0,130.6 and 186.2J/K - mol , respectively.

physical chemistry - Why doesn't Delta S total = 0 for this reversible process? - Chemistry Stack Exchange
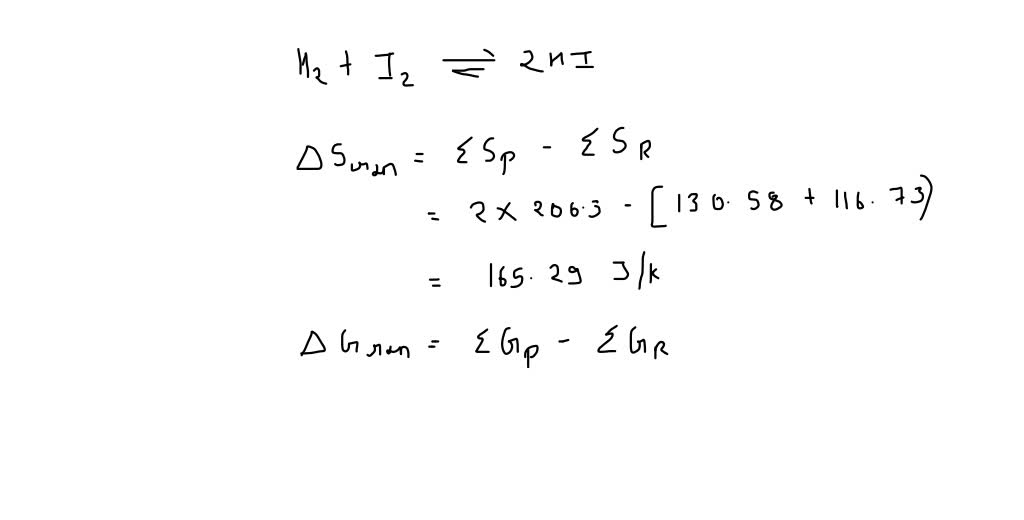
SOLVED: Use the values given below to Calculate delta S and delta H at 298K for the reaction H2 (g) + I2 (s) = 2HI (g) delta G delta S I2 (s)

OneClass: Can someone please calculate delta H and delta S from slope and intercept of the linear rel...
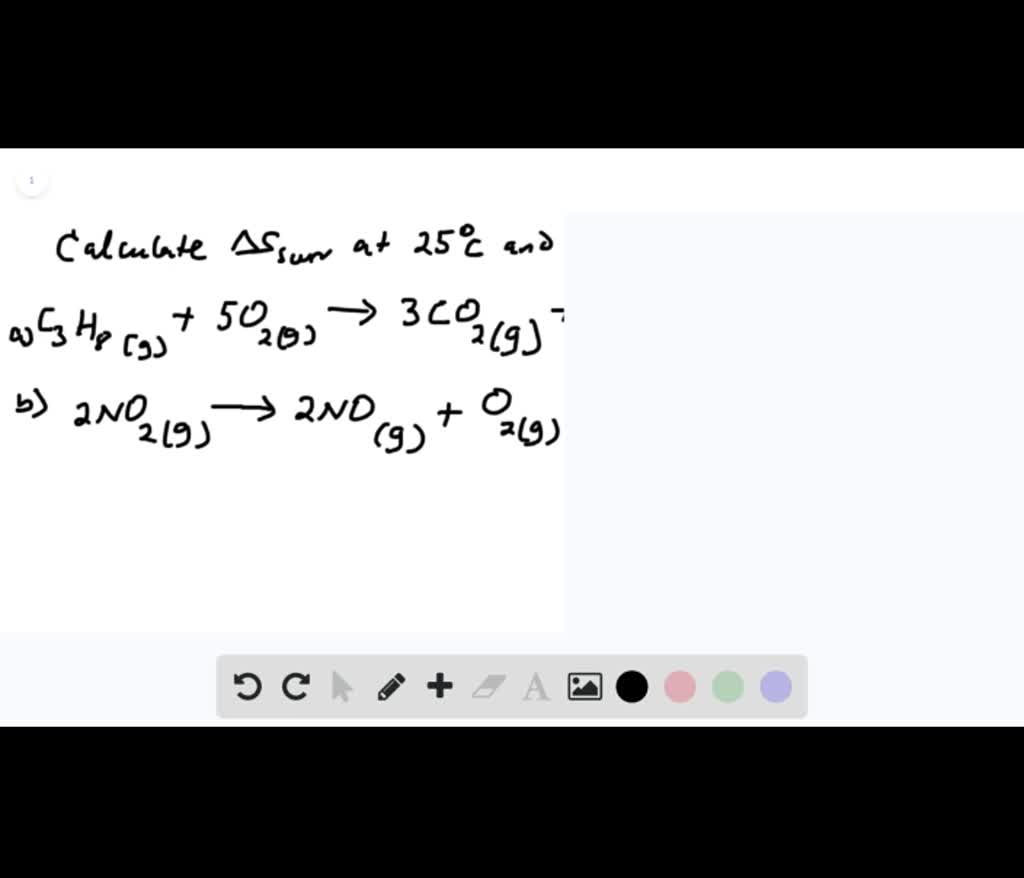
SOLVED:Calculate ΔS surr for the following reactions at 25^∘ C and 1 atm . a. C3 H8(g)+5 O2(g) ⟶3 CO2(g)+4 H2 O(l)ΔH^∘=-2221 kJ b. 2 NO2(g) ⟶2 NO(g)+O2(g) ΔH^ρ=112 kJ

DeltaH and DeltaS for the reaction Ag2O (s) iff 2Ag(s)+ 1/2O2(g) are 30.56 kJmol^-1 and 66.0 JK^-1 respectively. Calculate the temperature at which the reaction would be at equilibrium. Also predict the

SOLVED: For each reaction (a) predict the sign and then using the standard entropy tables (b) find the value of delta S P4 (s) + 5O2 (g) -> P4O10 (s) N5 (g) +