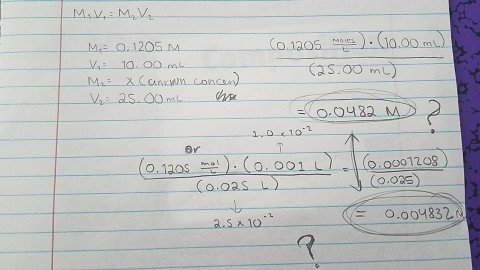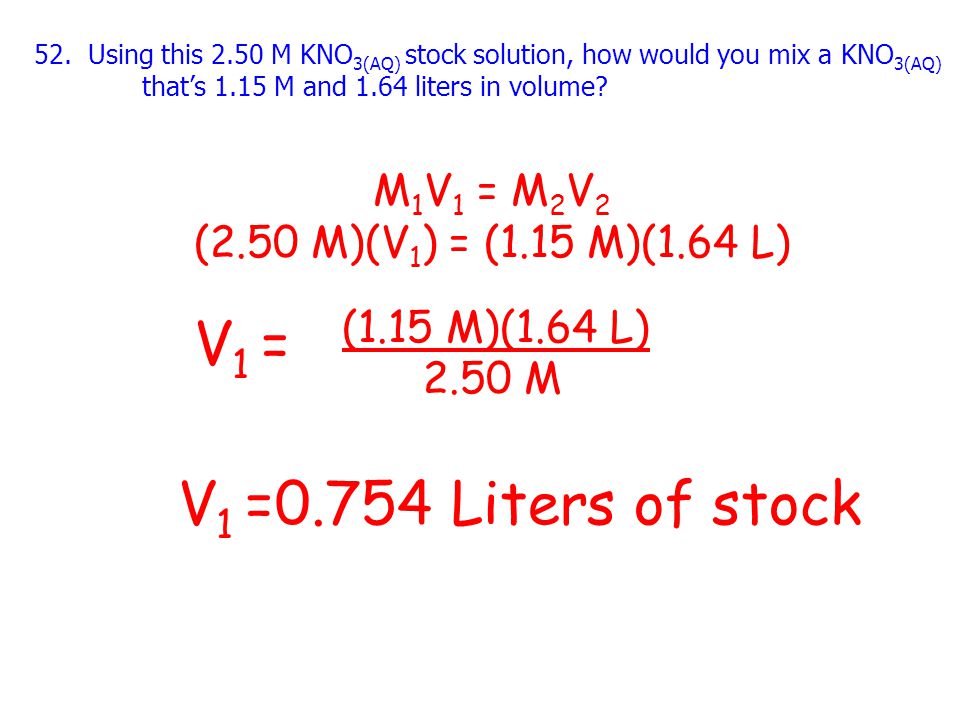
SOLUTIONS Class #1 Objective: Describing what solutions are, how they form, and how are they're strength is. - ppt download

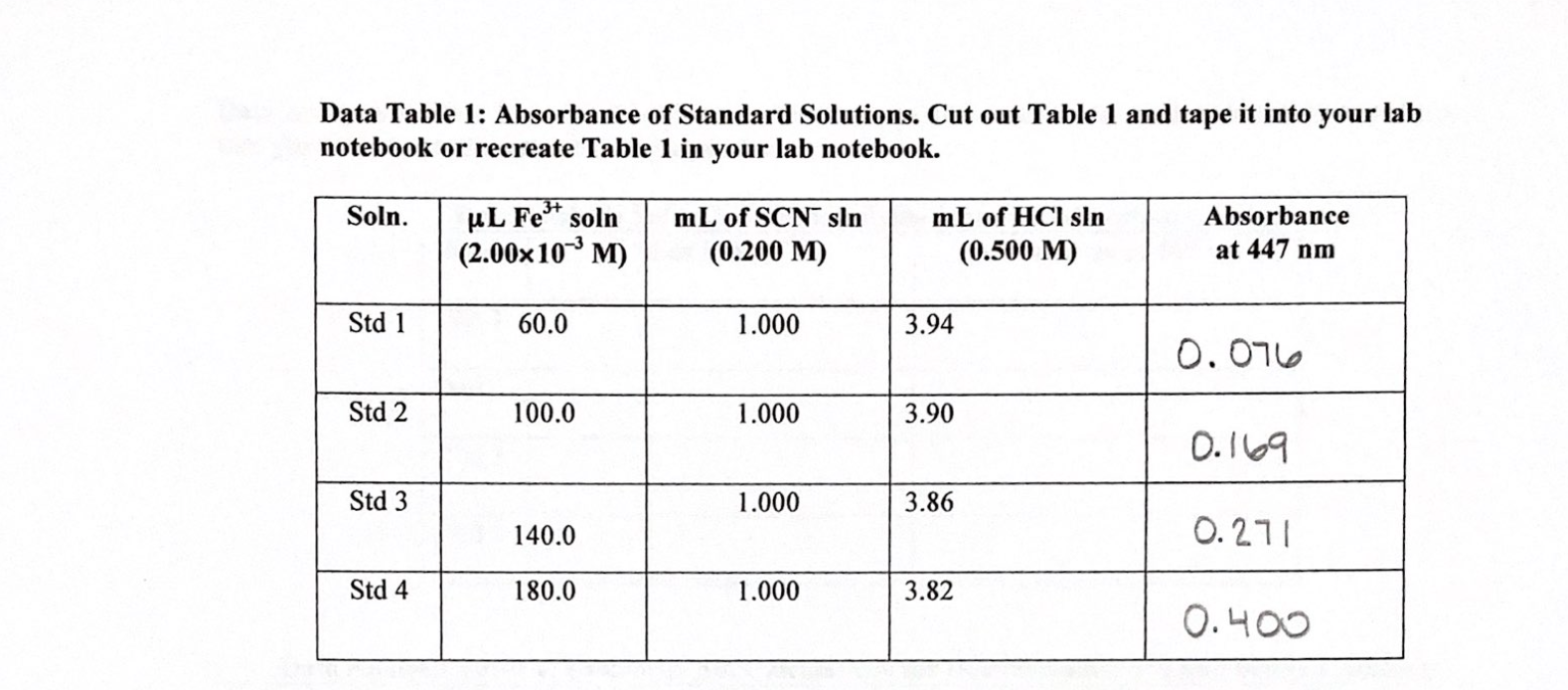
Using Spectrophotometry to calculate the mass of Copper in a sample of Brass – Shikhar's Lab Reports

SOLVED: Calculate the new molarity if each of the following dilutions is made. Assume the volumes are additive. (a) 52.3 mL of water is added to 23.3 mL of 0.123 M CaCl2