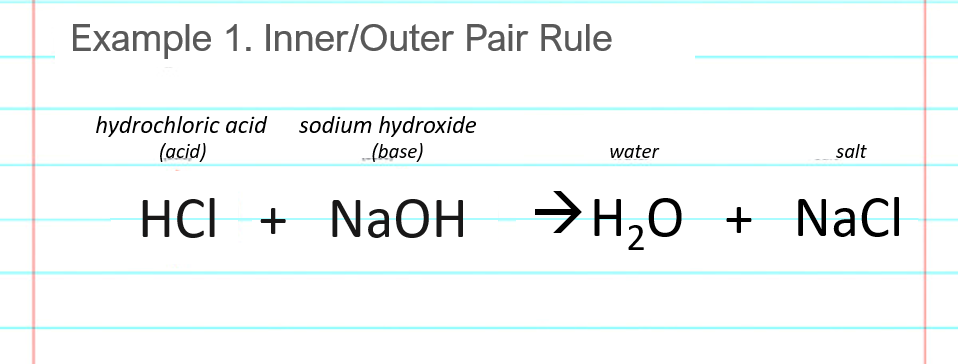
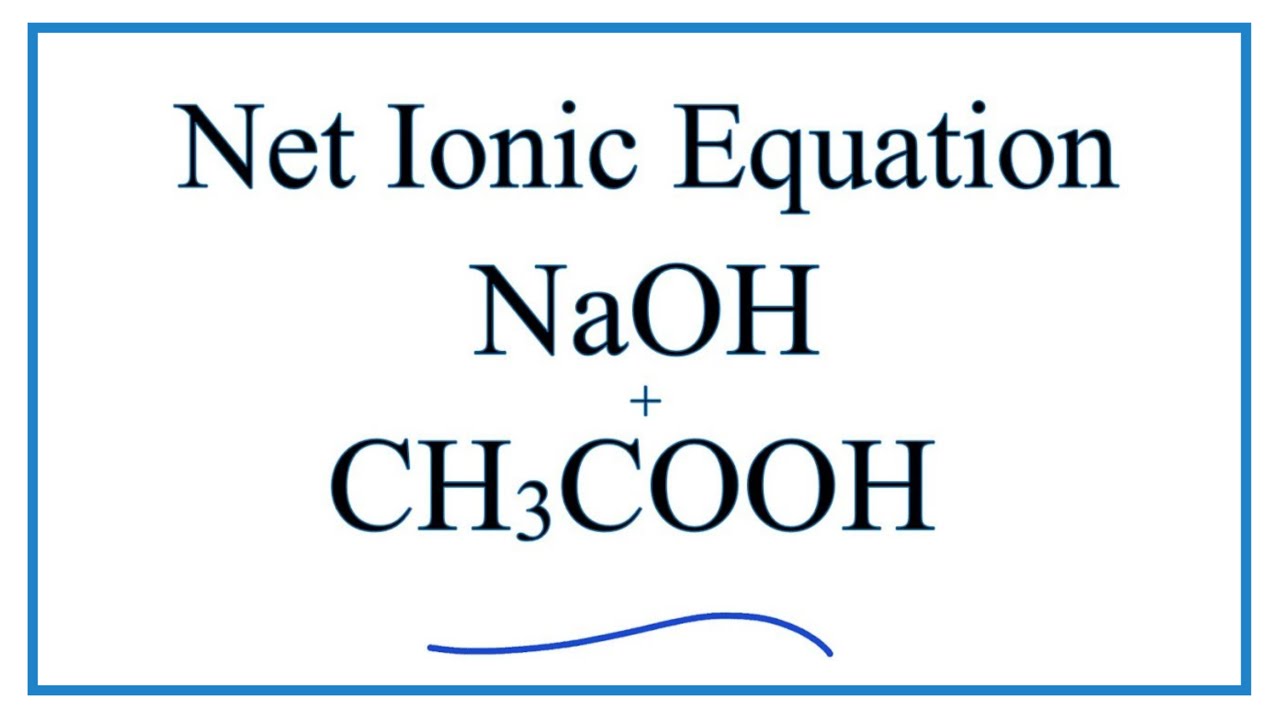Sodium hydroxide (NaOH) is classified as a strong base. For every mole of sodium hydroxide added to a large volume of water, one mole of what ion enters the solution? | Socratic
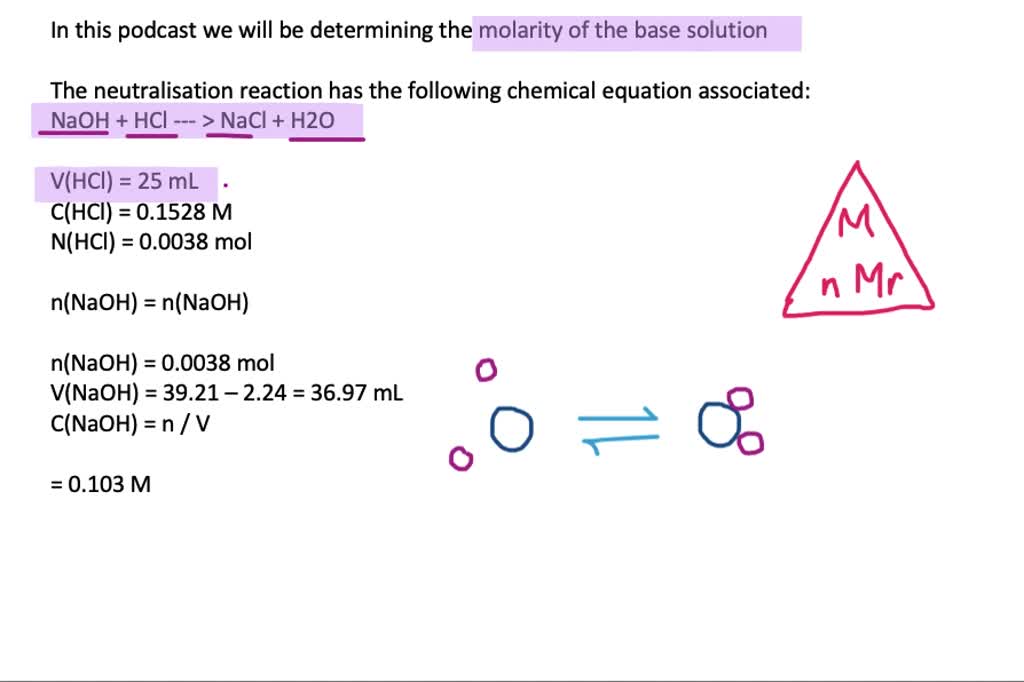
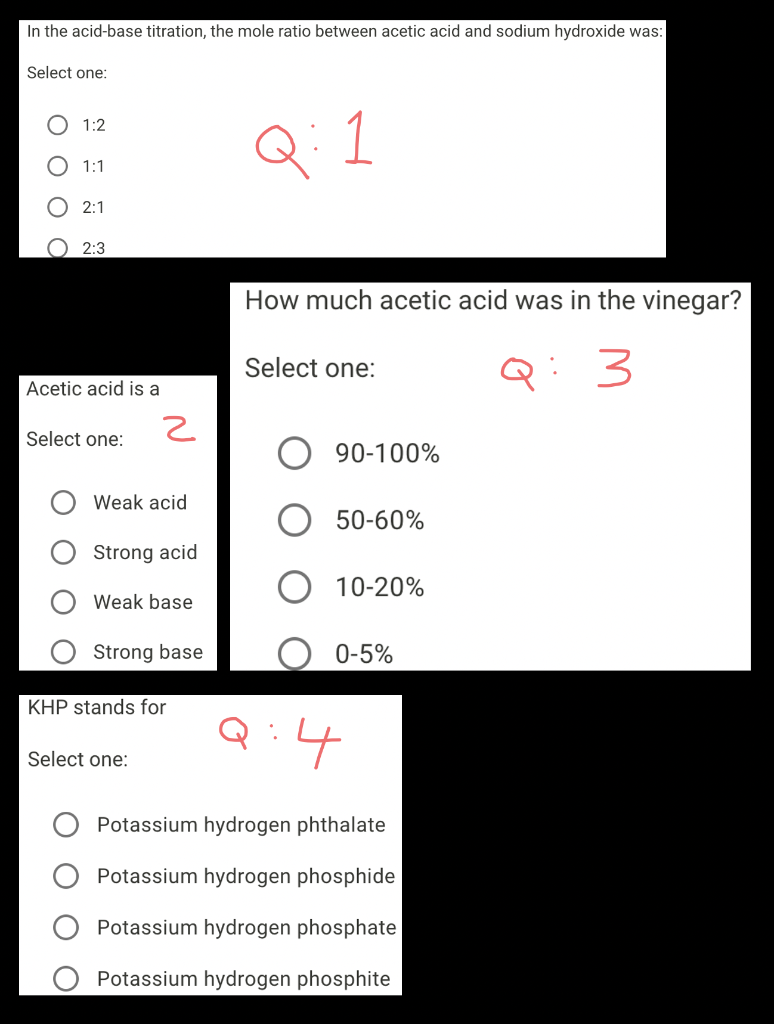
SOLVED:Sodium hydroxide is used extensively in acid-base titrations because it is a strong, inexpensive base. A sodium hydroxide solution was standardized by titrating 25.00 mL of 0.1528 M standard hydrochloric acid. The

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa
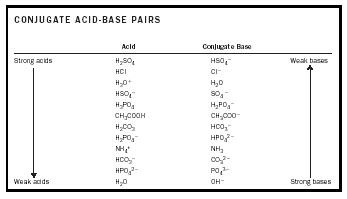
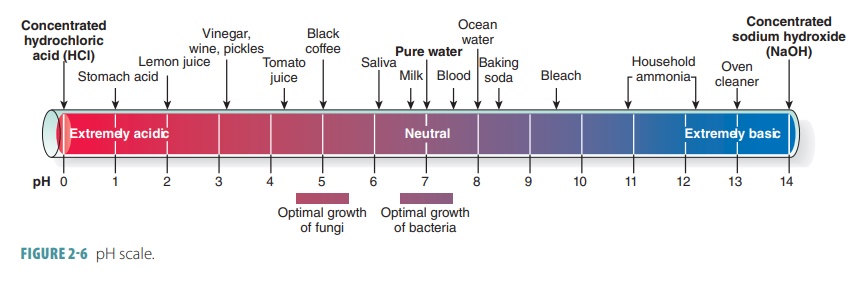
Acid-Base Chemistry - Chemistry Encyclopedia - reaction, water, metal, gas, number, equation, salt, property

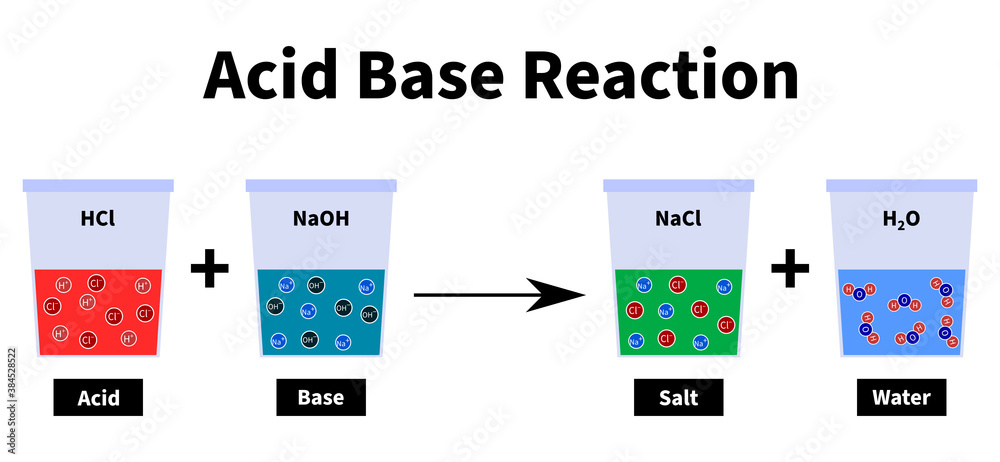
Acid – base reaction. chemical reaction neutralization the acid and base properties, producing a salt and water. used to determine pH. Bronsted – Lowry theory. molecules of HCl, NaOH, H2O, and NaCl,
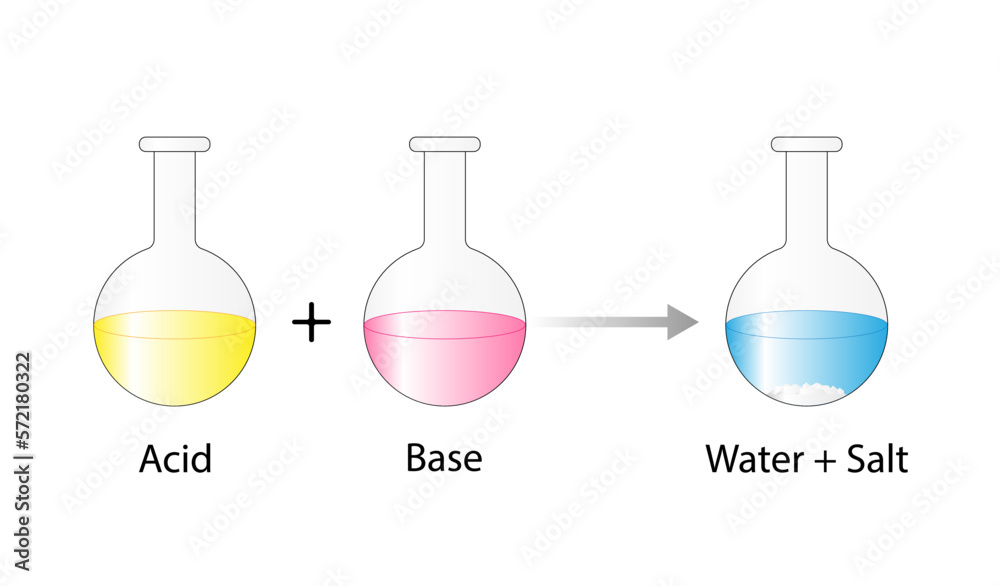
Acid–base reaction. chemical reaction neutralization. HCl hydrochloric acid, NaOH sodium hydroxide, and NaCl, sodium chloride. Vector illustration. Stock Vector | Adobe Stock

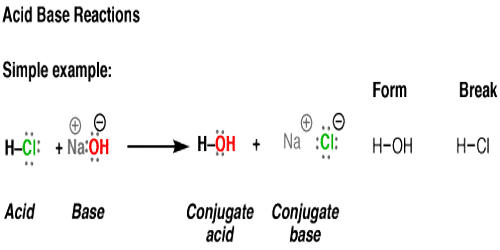
Write the neutralization reaction between Hydrochloric acid HCI and sodium hydroxide NaOH, and write the equation for this process.